
What is Density?
Teaching middle and high school students about density is paramount because it lays the groundwork for a deeper understanding of the physical world and fosters critical thinking. Density is all about how closely packed the particles in a substance are. It’s the ratio of mass to volume, and it’s everywhere around us, from sinking and floating objects to the delicious ice cream in our cones!
Density is the focus of this presentation, which is estimated to take approximately 20 minutes of class time. Guided notes are included for students to fill in while listening to and following along with the presentation.

Start with a Relatable Example: Ice Cream Cones
One of my favorite ways to introduce density is through ice cream cones. I mean, who doesn’t love ice cream? Imagine you have two ice cream cones—one with vanilla ice cream and the other with rocky road. Both cones have the same size (volume), but one is heavier than the other. Why? That’s where density comes in!
The vanilla ice cream has a lower density because it’s less dense than rocky road due to fewer solid particles and more air. The rocky road, on the other hand, has a higher density because it’s more densely packed with yummy chunks and less air. This tasty analogy helps students grasp the concept in a relatable way.
Analogies galore!
Analogies are a fantastic way to teach complex concepts like density by relating them to everyday experiences. Here are some engaging analogies to help your students grasp the idea of density:
- The Dance Floor Analogy: Imagine a crowded dance floor at a party. The number of people (mass) on the dance floor is like the density. If you have a small dance floor with many people, it’s dense, and they’re tightly packed. If you have a larger dance floor with the same number of people, it’s less dense, and there’s more space between them.
- The Fruit Basket Analogy: Think of a fruit basket filled with different fruits of varying sizes. The total weight of the fruits (mass) is the same, but the basket’s size (volume) determines how densely packed they are. A smaller basket is like a denser substance, while a larger basket is like a less dense one.
- The Traffic Analogy: Picture a busy road with cars. The number of cars on the road (mass) is like density. In heavy traffic, cars are closely packed, representing high density. In light traffic, cars are spaced farther apart, symbolizing low density.
- The Packed Suitcase Analogy: When preparing for a trip, packing your suitcase efficiently is key. If you stuff your suitcase full (mass) and it’s small (volume), it’s like a dense material. If you have the same amount of stuff but use a larger suitcase (volume), it’s like a less dense substance.
Hands-On Activities: Sink or Float?
Another engaging activity to teach density is the classic “Sink or Float” experiment. Gather a variety of objects like a cork, a stone, a plastic bottle cap, and a rubber ball. Challenge your students to predict whether each object will sink or float in a container of water. Then, let them test their hypotheses!
This activity allows students to apply their understanding of density. Objects that sink are denser than water, while those that float are less dense. It’s a hands-on way to reinforce the concept and encourage critical thinking.

Looking for an engaging and hands-on lab activity that will captivate your middle and high school students while teaching them the fundamental concept of density? Look no further! This “Mystery Density Lab” is the perfect addition to your classroom.
This lab is a fully immersive experience. Students get to roll up their sleeves and work with real-world materials, fostering a deeper understanding of mass, volume, density and critical thinking skills.
An answer key is included as well.
Density Tower: Building with Liquids
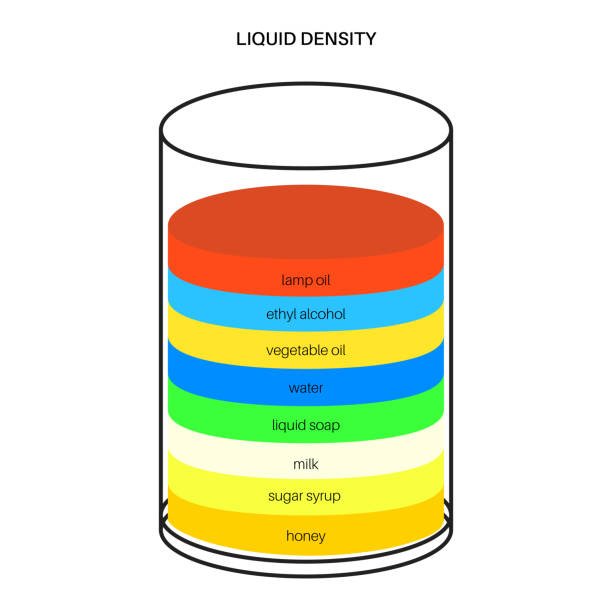
To take things up a notch, you can create a “Density Tower” with various liquids. Grab a clear container and carefully pour liquids like honey, dish soap, water, vegetable oil, and rubbing alcohol into it. Each liquid has a different density, so they’ll naturally layer according to their densities, creating a colorful and visually appealing tower.
As you pour each liquid, ask your students to predict the order of the layers based on density. This experiment not only showcases density but also sparks curiosity about why liquids don’t mix easily due to their varying densities.
Real-World Applications: Ship Design
To make the concept of density even more relatable, discuss real-world applications. For instance, talk about ship design. Ask your students why large ships made of steel can float on water despite being heavy. The answer lies in the ship’s shape and the principle of displacement, which relates to density.
Show them that a ship’s hull is designed to displace enough water to create an upward buoyant force that counters the ship’s weight. This application of density can really capture their imagination and show them the practical relevance of what they’re learning.
Final Words
Teaching density doesn’t have to be daunting. By using relatable examples, hands-on activities, and real-world applications, you can engage your students and make this fundamental concept both fun and memorable.

Looking for a full unit on density? Here’s a low-prep unit, which enables students to investigate the concept of density, how it is measured, and how it is applicable in our everyday lives.
Remember, the key is to foster curiosity and encourage questions. Science is all about discovering the world around us, and density is just one exciting piece of the puzzle.
















